1932
Began production and sale of sodium silicate in Osaka under the company name Takahashi Water Glass Manufacturer.
1951
Established Fuji Sodium Silicate Co., Ltd.. in Tokyo and began producing silica gel: began nationwide sales in Japan; continued to produce and sell sodium silicate.
1957
Established Kozoji Chemical Industrial Co., Ltd., (predecessor of the present Kasugai Plant) and began specialized production of silica gel.
1961
Fuji Sodium Silicate Co. Ltd., Meika Industrial Co., Ltd., and Takahashi Water Glass Manufacturing Co., Ltd., merged to form Fuji Chemical Co.. Ltd. Fuji Chemical Co., Ltd., acquired Keiso Chemical Laboratory.
1962
Established a plant for the production of silica gel in Honai-cho. Nishiuwa-gun, Ehime Prefecture (the present Ehime Plant].
1965
Split the sodium silicate operations and silica gel operations of Fuji Chemical Co., Ltd., and transferred the business rights for the silica gel operations to Yugen Kaisha Fuji.
1965
Fuji-Davison Chemical Ltd. established through 50% capital investment each by the Davison Chemical Division of W.R. Grace & Co. and Yugen Kaisha Fuji.
1966
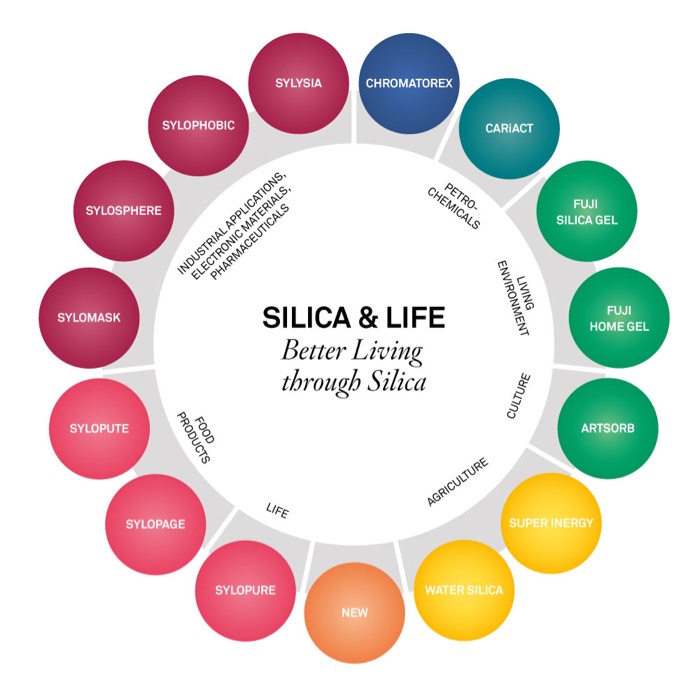
Started production and sale of Syloid.
1973
Compleled construction ol Head Office building in Kozoji-cho, Kasugai City, Aichi Prefecture; opened Tokyo Sale\ Office.
1978
Opened Hiroshima Sales Office and Fukuoka Sales Office.
1979
Opened Nagoya Sales Office.
1984
Compleled Hyuga Plant.
1986
Completed Technical Center; opened Osaka Sales Office and South Korea Branch Office.
1989
First phase expansion of Hyuga Plant.
1992
Second phase expansion of Hyuga Plant.
1993
Dissolved joint venture with W.R. Grace & Co. and changed company name to Fuji Silysia Chemical, Ltd.; opened U.S. Branch Office.
1998
Certification under ISO 9002 (production of micronized silica products at Kasugai Head Office Plant and Hyuga Plant); established Fuji Silysia Chemical USA. Ltd.
1999
Third phase expansion of Hyuga Plant; established Fuji Silysia Chemical S.A. in Switzerland; closed Hiroshima Sales Office (operations transferred to Osaka Sales Office and Fukuoka Sales Office for consolidation of sales office operations).
2000
Established Silysiamont S.p.A. in ltaly as a joint venture with Ausimont S.p.A. (now Solvay S.A.); U.S. plant completed in Greenville, North Caroline, by Fuji Silysia Chemical USA. Ltd.; additional certification under ISO 9002 (production of spherical powder silica gel products at Kasugai Head Office Plant); closed South Korea Branch Office.
2001
Certification changed from ISO 9002 to ISO 9001.
2002
Certification under ISO 14001 (Hyuga Plant); completed Italy Plant at Silysiamont S.p.A.
2003
Additional certification under ISO 9001 (production of spherical powder silica gel products at Hyuga Plant).
2005
Certification under lSO 14001 (Kasugai Head Office Plant). Fourth phase expansion of Hyuga Plant.
2006
Additional certification under ISO 9001 (production of spherical silica gel products at Ehime Plant).
2011
Established liaison office in Phillippines.
2012
Relocations of sales offices in Tokyo and Nagoya.
2013
Relocation of sales office in Osaka.
2014
Established KYRAPHARM SAS in France as a joint venture with AXYNTIS SAS.
2015
Invest in 50% of AXYNTIS
2016
Silysiamont S.p.A. in ltaly 100% Fuji Silysia Chemical Ltd.
2017
Established Silylab plant at Pithiviers, France
2023
Dissolved joint venture of KYRAPHARM SAS with AXYNTIS SAS.